
MCAL Therapeutics traces its origin to a question sparked by nature itself. Dr. Daniel Albert, a world-renowned ophthalmologist and member of MCAL’s Board of Directors, had long been fascinated by a biological fact: rabbits blink only four times per hour and can sleep with their eyes open – but do not suffer from dry eye. In contrast, humans blink approximately 840 times per hour (about 14 times per minute).
What protected their eyes so effectively? Driven by curiosity and decades of clinical insight, MCAL Therapeutics hypothesized that evolutionary biology might hold the key to rethinking dry eye therapy altogether. The answer lay in the rabbit’s unusually stable tear film – a hyper-stabilized lipid layer that resists evaporation and maintains ocular moisture far better than the human equivalent. That insight ignited the founding vision behind MCAL: if nature had already solved the challenge of a hyper-stabilized tear film through lipid biology, why not produce a synthetic version for human use?
Thus began the journey to create MCAL-201, a rabbit-inspired, slow-release, synthetically produced lipid eye drop designed to restore the tear film’s protective barrier, offering a radically new approach to treating evaporative dry eye disease.
Team
With a leadership team and Board bringing many decades of experience advancing ophthalmic innovations that have transformed patient care – including first-in-class and category-defining therapies such as Xiidra®, Lucentis®, and EYLEA® – MCAL Therapeutics is applying deep translational expertise to develop scalable, science-driven solutions for patients living with evaporative dry eye and other ophthalmic diseases.
LEADERSHIP TEAM



BOARD OF DIRECTORS
Science
Understanding Evaporative Dry Eye
Evaporative Dry Eye (EDE) is the most common – and least treated – form of dry eye disease, affecting an estimated 18-24 million Americans.
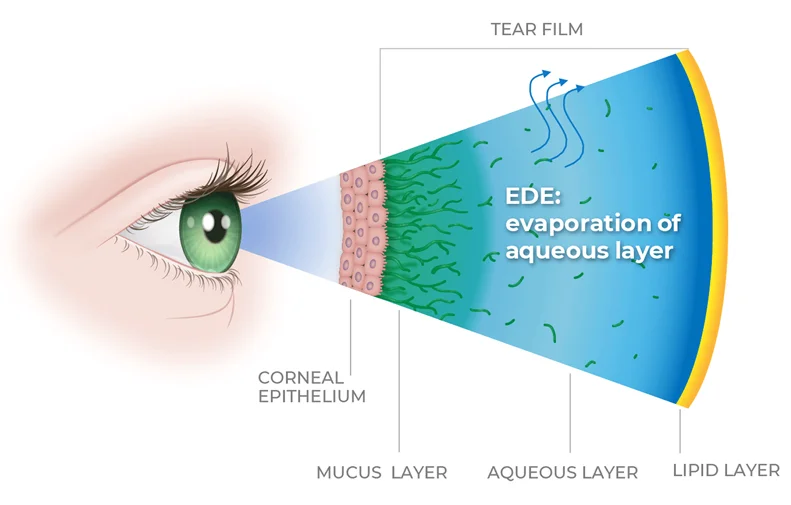
Unlike Aqueous Deficient Dry Eye (ADDE), which results from reduced tear production, EDE stems from a breakdown in the tear film’s lipid layer often due to meibomian gland dysfunction. This lipid layer is essential for preventing tear evaporation and maintaining a healthy ocular surface.
Patients with EDE experience:
- Dryness, burning, stinging, and blurred vision
- Swollen eyelids and crusting
- Frequent blinking
- The sensation of “sand in the eyes”
While prescription therapies for ADDE aim to reduce inflammation, they do not directly address the root cause of EDE – tear film instability and lipid deficiency.
MCAL-201 is designed to fill this critical gap, with a novel, synthetic lipid eye drop designed to restore tear film structure and gland function for relief that lasts.
Pipeline
A Versatile Product. A Singular Focus on Lasting Relief.
MCAL-201 is our lead therapy, developed from MCAL’s proprietary crystalline lipid suspension platform – a scalable, extended-release delivery system designed for topical application across ocular and other mucosal diseases. Stable at room temperature, the platform redefines how lipid-based therapies are formulated and delivered, supporting once-daily dosing. In preclinical studies, the system has demonstrated sustained release of co-formulated agents for topical use in diseases of the eye and other mucosal surfaces.
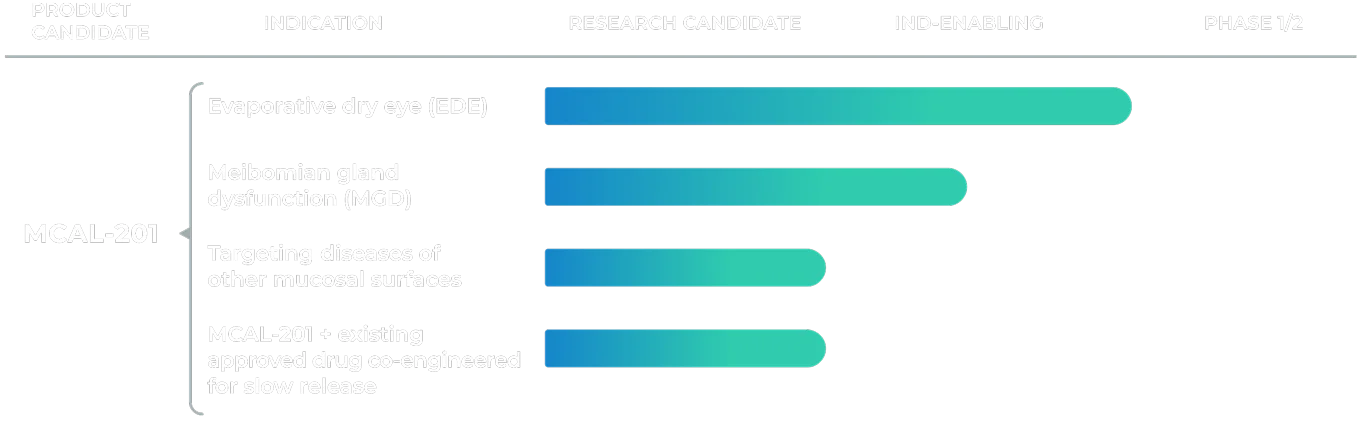
MCAL-201 for Evaporative Dry Eye
Our first program targets evaporative dry eye – the most common, least treated form of dry eye disease. Preclinical studies demonstrated restoration of tear film function, including improved tear film breakup time (how long the protective tear film on the surface of your eye remains stable and intact after a blink), enhanced stability, and recovery of meibomian gland performance. We plan to initiate a Phase 1/2a clinical trial during the second half of 2026.
The Science Behind the Pipeline
MCAL’s crystalline lipid system is designed for flexibility, scalability, and long-lasting relief – today in dry eye, tomorrow across other ocular and mucosal diseases.
- 99% natural ether lipid
- Micronized crystalline suspension for durable delivery
- 6 months of chemical stability at room temperature
- Potential for co-engineered formulations targeting other ocular and mucosal surface disorders
